 The negative electrons are pulled strongly enough by the positively charged anode that they whack the electrons in the O2 and N2 molecules, putting them into higher energy states or knocking them free of the molecule. The light emitted from the electron stream comes from nitrogen and oxygen molecules. 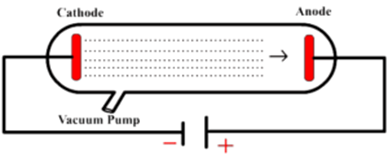
I know the questions are very silly but because different websites refer to different things, I am becoming confused with something that should be simple to understand. Could someone please tell me why these conditions were necessary? For example, " The cathode rays consist of material particles because they produced shadow of objects placed in the way"ģ.Two of the conditions of the experiment were air at very low pressure and secondly a very high potential difference. The path cathode rays travel is not affected by the position of the anode." I just can't seem to understand this explanation of the one of the observations.Also, different websites analyses this observation differently. That is why, cathode rays cast shadow of any solid object placed in their path. Is this the color of the radiation itself?Ģ." Cathode rays travel in straight lines. However, as shown in the above diagram there was no fluorescent material in the experiment carried out first on the cathode ray tube. Many websites I read through refer to a fluorescent material. Pierre Curie carried a vial of radium in his coat pocket to demonstrate its greenish glow, a habit that caused him to become ill from radiation poisoning well before he was run over by a horse-drawn wagon and killed instantly in 1906.1.One of the observations I learned was that the glass tube begins to glow with a brilliant green light. Starting with several tons of pitchblende, the Curies isolated two new radioactive elements after months of work: polonium, which was named for Marie’s native Poland, and radium, which was named for its intense radioactivity. She found that one particular uranium ore, pitchblende, was substantially more radioactive than most, which suggested that it contained one or more highly radioactive impurities. Marie Curie coined the term radioactivity (from the Latin radius, meaning “ray”) to describe the emission of energy rays by matter. Becquerel’s work was greatly extended by Marie Curie (1867–1934) and her husband, Pierre (1854–1906) all three shared the Nobel Prize in Physics in 1903. 
The second line of investigation began in 1896, when the French physicist Henri Becquerel (1852–1908) discovered that certain minerals, such as uranium salts, emitted a new form of energy. With this information and Thomson’s mass-to-charge ratio, Millikan determined the mass of an electron: Subsequently, the American scientist Robert Millikan (1868–1953) carried out a series of experiments using electrically charged oil droplets, which allowed him to calculate the charge on a single electron. 
Another set of electrode plates deflect the ray, with the ray bending towards the positive plate. Schematic of cathode ray tube with deflection. Image used with Permission (CC BY-SA-NC). As the cathode rays travel toward the right, they are deflected toward the positive electrode (+), demonstrating that they are negatively charged. \): Deflection of Cathode Rays by an Electric Field. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
